Your Location:Home > Products > Pharmaceutical > THCV
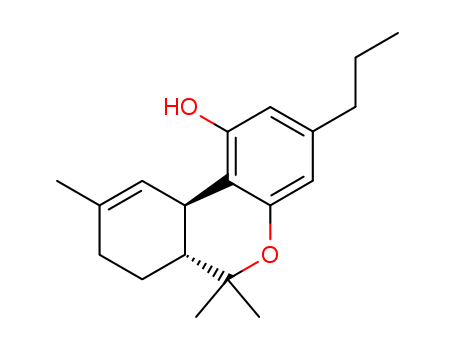

CasNo: 31262-37-0
Molecular Formula: C19H26 O2
|
31262-37-0 Name |
|
|
Name |
THCV |
|
Synonym |
Δ9-Tetrahydrocannabivarin;Δ9-Tetrahydrocannabivarol;Δ9-trans-Tetrahydrocannabivarin;Tetrahydrocannabivarin (CRM);Delta-9-tetrahydrocannabivarin (THCV);Tetrahydrocannabivarin;THCV (Tetrahydrocannavarin) 99%;(6aR)-3-Propyl-6,6,9-trimethyl-6aβ,7,8,10aα-tetrahydro-6H-dibenzo[b,d]pyran-1-ol |
|
31262-37-0 Chemical & Physical Properties |
|
|
Boiling point |
360.6±42.0 °C at 760 mmHg |
|
Density |
1.0±0.1 g/cm3 |
|
Molecular Formula |
C19H26O2 |
|
Molecular Weight |
286.409 |
|
Flash Point |
137.6±22.1 °C |
|
PSA |
29.46000 |
|
LogP |
6.62 |
|
Exact Mass |
286.193268 |
|
Vapour Pressure |
0.0±0.8 mmHg at 25°C |
|
Index of Refraction |
1.536 |
THCV is a naturally occurring analog of THC. Δ9-Tetrahydrocannabivarin (THCV) is a cannabis-derived compound with unique properties that set it apart from the more common cannabinoids, such as Δ9-tetrahydrocannabinol (THC). The effect of THCV on dyslipidemia and glycemic control in type 2 diabetics showed reduced fasting plasma glucose concentration when compared to a placebo group.
InChI:InChI=1/C19H26O2/c1-5-6-13-10-16(20)18-14-9-12(2)7-8-15(14)19(3,4)21-17(18)11-13/h9-11,14-15,20H,5-8H2,1-4H3/t14-,15-/m1/s1
This work paves the way for the development of new therapeutic means based on THCV and its regenerative properties to create a favorable environment for the development of healthy mature adipocyte tissue and to reduce the incidence and clinical outcome of metabolic diseases such as diabetes. We also observed that different concentrations of THCV in the range of 0–10 μM did not affect either cell viability or population doubling time (Figure 1B,C); however, THCV used at 50 and 100 μM reduced cell viability by up to 40%.
THCV significantly decreased fasting plasma glucose (from 7.4 to 6.7 mmol/L) compared to the placebo group which increased from 7.6 to 8 mmol/L 21 with an estimated treatment difference (ETD) of − 1.2 mmol//L, p < 0.05. In contrast to THC, clinical and therapeutic advantages of THCV regarding its lack of psychoactive effects in human studies are of great value in pharmacotherapy.
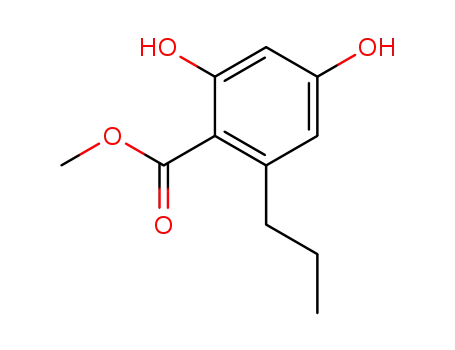
2,4-dihydroxy-6-n-propylbenzoic acid, methyl ester

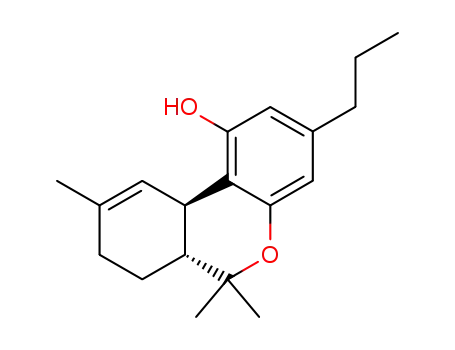
tetrahydrocannabivarin
| Conditions | Yield |
|---|---|
|
Multi-step reaction with 4 steps
1.1: boron trifluoride diethyl etherate / toluene / 20 °C
1.2: 0.42 h / 20 °C
2.1: potassium hydroxide / 2 h / 100 - 120 °C / 375.04 Torr
3.1: 150 °C
4.1: boron trifluoride diethyl etherate / dichloromethane / 0.5 h / 0.22 °C
With boron trifluoride diethyl etherate; potassium hydroxide; In dichloromethane; toluene;
|
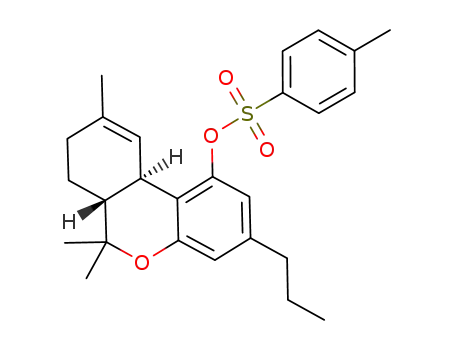
Δ9-tetrahydrocannabivarin tosylate


tetrahydrocannabivarin
| Conditions | Yield |
|---|---|
|
With potassium tert-butylate; water; In tert-butyl alcohol; at 65 ℃; for 4.5h;
|
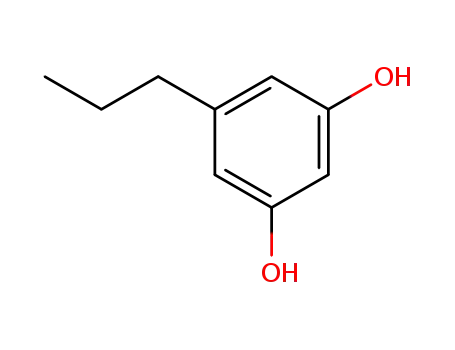
5-propyl-1,3-benzenediol
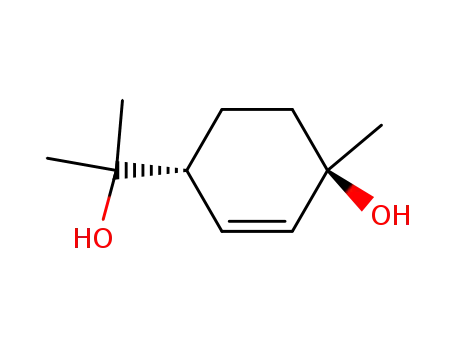
cis-Δ2-p-menthene-1,8-diol

Δ9-tetrahydrocannabivarin tosylate

2,4-dihydroxy-6-n-propylbenzoic acid, methyl ester

Δ9-tetrahydrocannabivarin tosylate
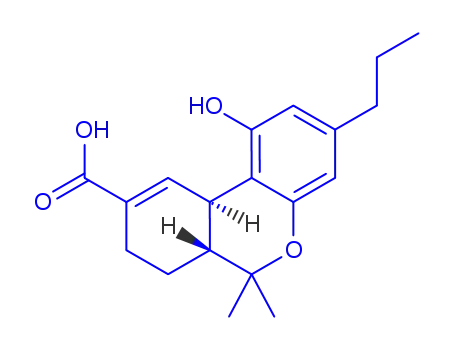
Δ9-tetrahydrocannabivarin-11-oic acid
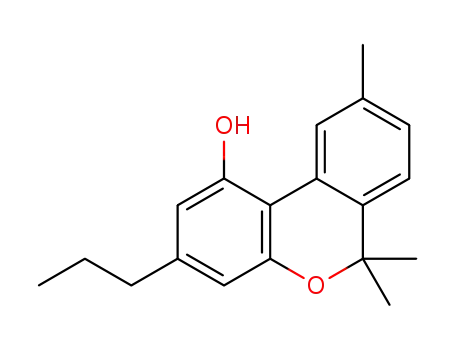
cannabivarin