Your Location:Home > Products > Food Additives > Silica gel, pptd., cryst.-free


CasNo: 112926-00-8
Molecular Formula: O2Si
Appearance: white fluffy powder
|
112926-00-8 Name |
|
|
Name |
Silica Gel |
|
Synonym |
Silica gel GF Pre-coated Plate for TLC;High Silica gel HF254 Pre-coated Plate for TLC;Silica gel H Pre-coated Plate for TLC;AEROSIL(TM) 200;Silica gel G Pre-coated Plate for TLC;High Silica gel GF Pre-coated Plate for TLC;silica,amorphous,precipitatedandgel;silicagel,pptd.,cryst.-free |
|
112926-00-8 Chemical & Physical Properties |
|
|
Melting point |
1610°C |
|
Boiling point |
2230°C |
|
Density |
2.6 |
|
Molecular Formula |
(SiO2)x(H2O)y |
|
Molecular Weight |
60.0800018310547 |
|
Flash Point |
23°C |
|
Appearance of Characters |
blue |
|
Storage condition |
2-8°C |
|
PH |
~6.8 (10% in aq. suspension) |
|
Water Solubility |
Insoluble in water |
Silica gel, an inorganic polymer, is well-known due to its application in chromatographic columns. Its porous surface structure and large specific surface area of above 700 m2 g−1 play important roles for its interaction with metal ions. In addition, as a matrix for immobilizing different chemical functional groups, silica gel is readily accessible.
Silica gel is a rigid 3D network of colloidal silica. It is exclusively used as a support material for the active titanium(III) centers in Ziegler-Natta catalysts. Silica gel was used on glass plates for preparative TLC and on aluminium sheets for analytical TLC. A few studies model the silica gel bed as loose grains packed in between the heat exchanger tubes/fins and the vapor is assumed to be stagnant. The effects of the silica gel on the thermal conductivity and strength of the composite were investigated. Silica gel has also been used to encapsulate certain pollutants and solid wastes due to its high physical strength, durability, and chemical stability.
For silica gels, a higher capacity for water adsorption after 5000 thermal swing adsorption cycles was observed in samples with a lower concentration of surface silanol groups. The results from TGA analysis of the silica gels showed that the concentration of silanol groups on the 2.2 nm pore size silica gel was 2.5 OH nm−2, the 3.0 nm pore size had 2.3 OH nm−2, and the 6.0 nm pore size had 2.1 OH nm−2.
In parallel, the synthesis of a modified silica gel carrier (3-azidopropyl-functionalized silica gel) via a 3-chloropropyl intermediate was carried out. The pre-functionalized silica gel matrices were characterized by both solid-state 29Si and 13C NMR as well as EA. Attempts to increase the loading amount by increasing the number of anchor points (alkyne-groups) on the CS and azido-groups on silica gel were made but were unsuccessful given the low accessible surface area of the silica material.
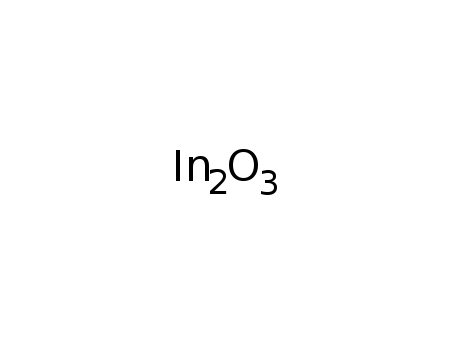
indium(III) oxide

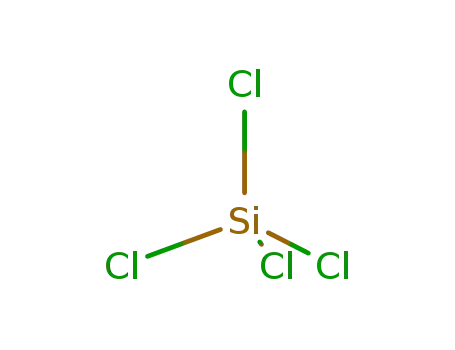
tetrachlorosilane


silica gel

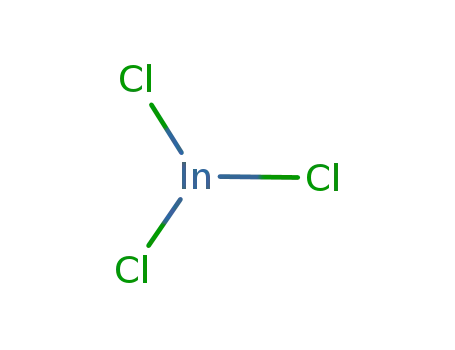
indium(III) chloride
| Conditions | Yield |
|---|---|
|
780-800°C in vac.; Two-fold vacuum sublimation with prior distilling off SiCl4.;
|

tetrachlorosilane

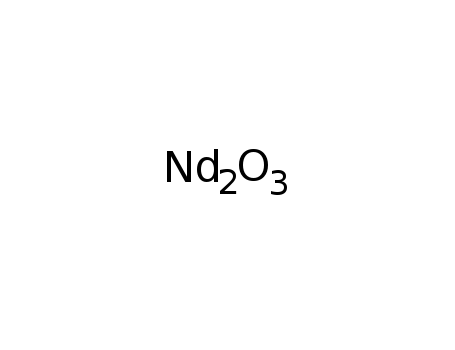
neodymium(III) oxide

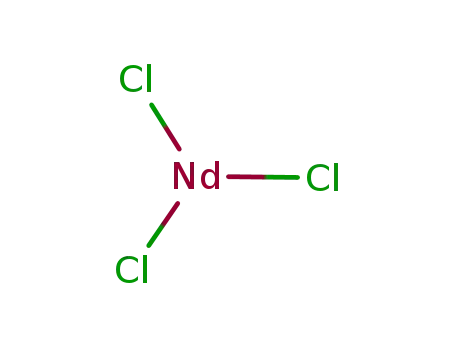
neodymium trichloride


silica gel
| Conditions | Yield |
|---|---|
|
1000°C in vac.; distilling;
|

tetrachlorosilane
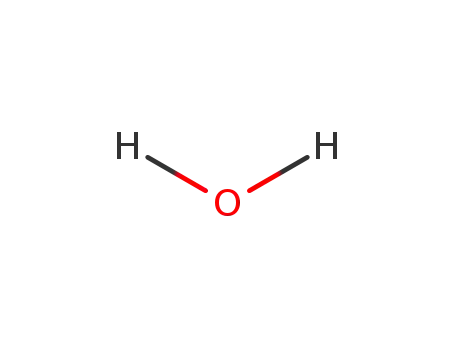
water
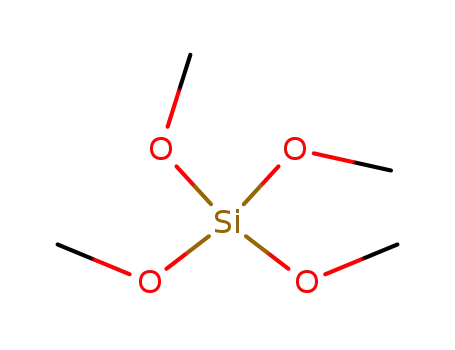
tetramethylorthosilicate

ethenyltrimethylsilane
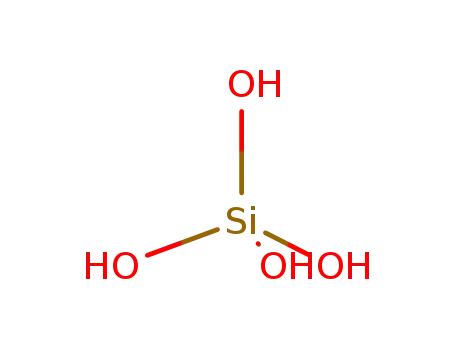
silicic Acid
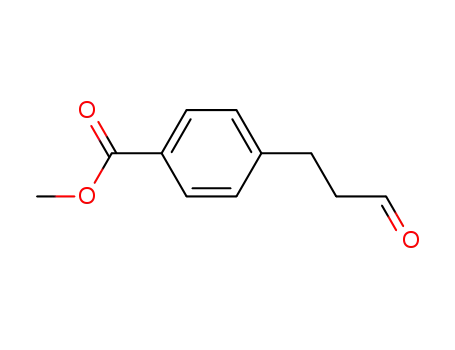
4-(3-oxo-propyl)-benzoic acid methyl ester
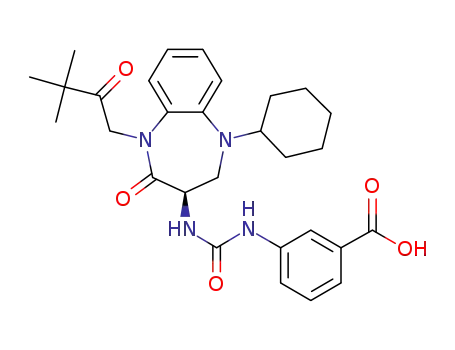
(R)-(-)-3-[3-(1-tert-butylcarbonylmethyl-2-oxo-5-cyclohexyl-1,3,4,5-tetrahydro-2H-1,5-benzodiazepin-3-yl]ureido]benzoic acid
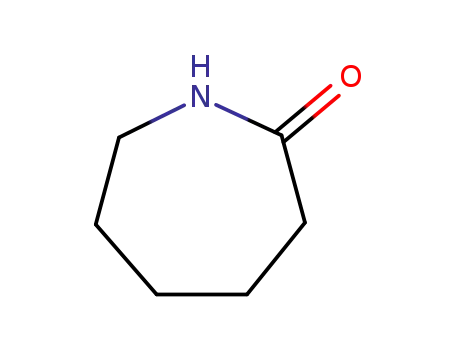
caprolactam